Blog
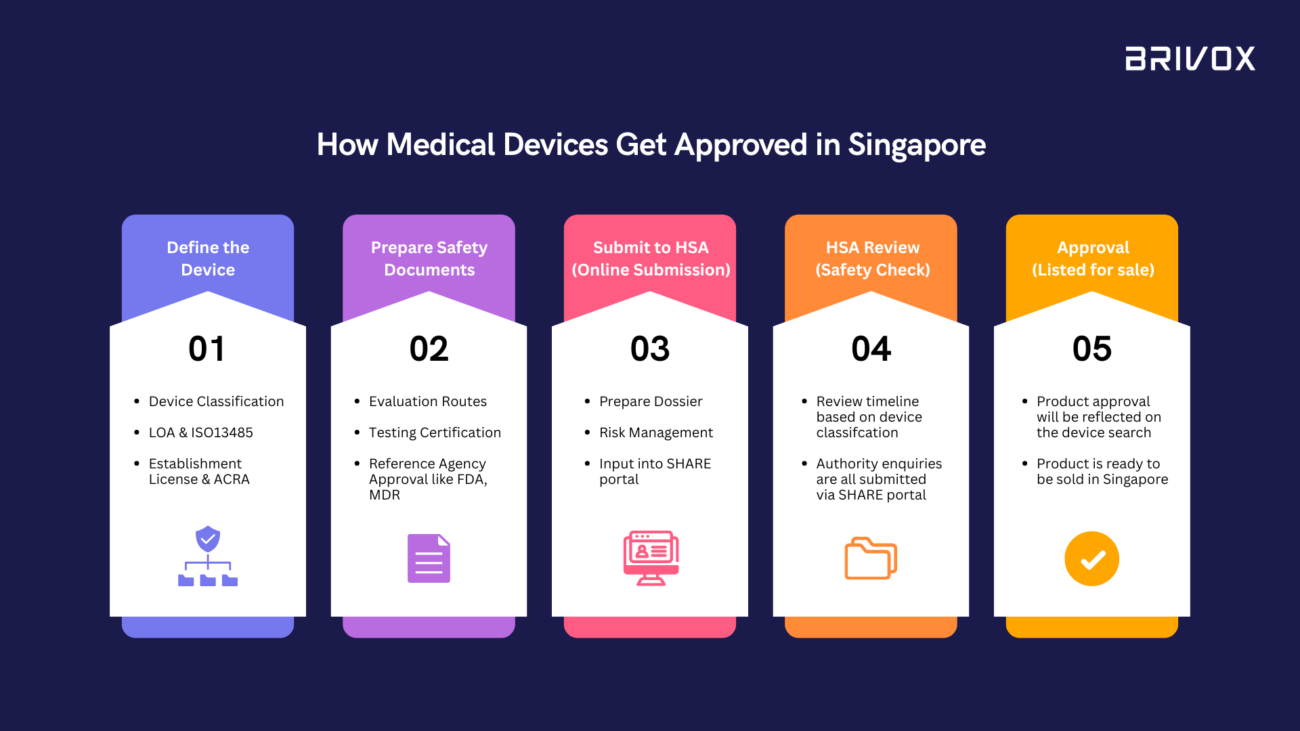
Bringing a medical device to market in Singapore involves more than just innovation. Every device must pass through a structured regulatory process overseen by the Health Sciences Authority (HSA) to ensure safety, quality, and effectiveness. For many...
At Brivox, we understand that healthcare and medical device distribution is not just about products—it’s about trust, safety, and consistent quality. That’s why our operations are guided by internationally recognized standards, and why we proudly...
One of the most common questions from medical device companies is:“How long will it take to get approval in Singapore?” The short answer: it depends on your device classification, documentation, and regulatory strategy.The long answer? Let’s break it...