How Medical Devices Get Approved in Singapore: A Simple Guide for Innovators
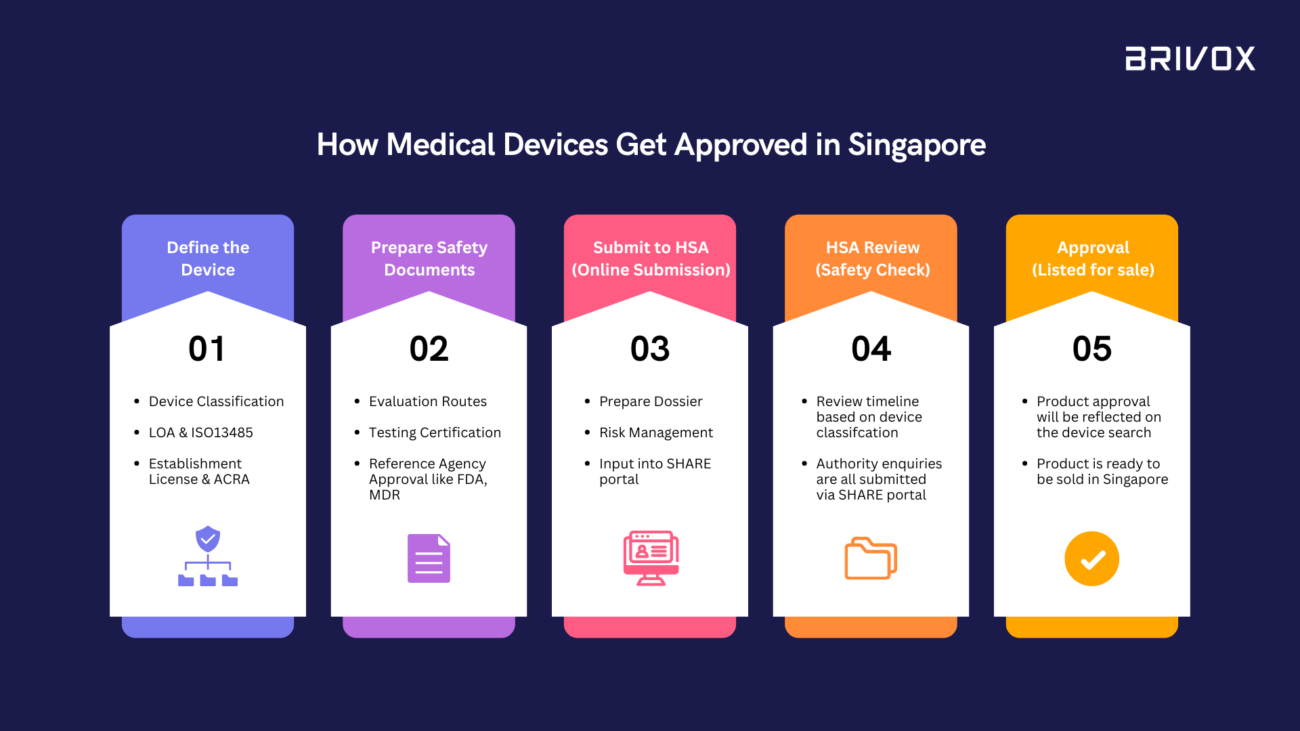
Bringing a medical device to market in Singapore involves more than just innovation. Every device must pass through a structured regulatory process overseen by the Health Sciences Authority (HSA) to ensure safety, quality, and effectiveness.
For many startups and manufacturers, navigating this process can feel complex. Understanding the key stages can help you prepare early and accelerate market entry.
Below is a simplified overview of how medical devices are approved in Singapore.
Brivox’s Strengths
Brivox supports medical device manufacturers with strong regulatory knowledge and market experience.
- ISO 13485 certified by the BSI Group
- Strong domestic market understanding
- Assistance with OEM processes and product actualisation
- Certified Lead Auditor expertise in device and material consultancy
- Sterilisation protocol, process and consultancy
- Distributorships with robust market channels
- Strong training and sales team
What Brivox Can Offer
Brivox helps innovators move from development to commercialisation.
- Product registration
- Dossier and licensing consultancy
- Product distributorship with strong market presence
- Support for Asia market penetration and distribution
- Co-develop, implement, actualise and commercialise devices
- Link agencies, institutions, centres and doctors for product innovation
- A ready manufacturer and distributor for device launch